Fluoride poisoning from Veozah Fezolinetant?
Trying to find the metabolites of Fluorinated drugs in Humans is tricky.
Menopausal women seeking non-hormonal relief from hot flushes have been reporting worrying symptoms, including Heart Attack (4 cases in FAERS), after taking tablets of a Fluorinated drug Fezolinetant, also known as Veozah or ESN364, prompting a recent US FDA warning.12
Ingredients
A friend on Gettr pointed me to the ingredients:
Each VEOZAH (fezolinetant) tablet for oral use contains 45 mg of fezolinetant and the following inactive ingredients: ferric oxide, hydroxypropyl cellulose, hypromellose, low-substituted hydroxypropyl cellulose, magnesium stearate, mannitol, microcrystalline cellulose, polyethylene glycol, talc, and titanium dioxide.
No Information at US CTD
I naturally turned to the US Government Comparative Toxicogenomics Database and found the cupboard is bare.3 Click to expand.
Perhaps Robert F Kennedy can request curation if he is part of the next US President’s team.
Defluorination of ESN364?
Previously I mentioned Defluorination of drugs incorporating a Fluorine atom on an aromatic ring in various drugs, yielding free toxic Fluoride ions.4
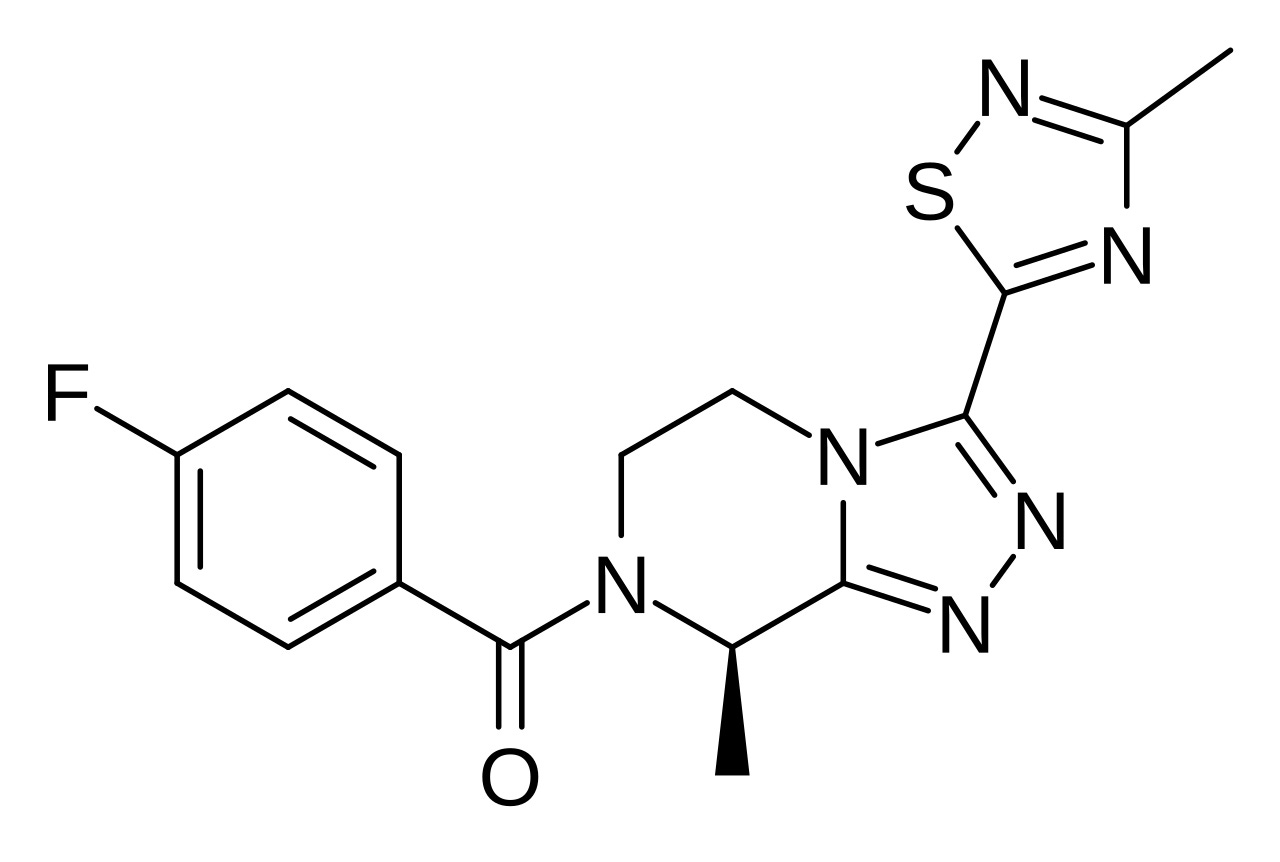
Here is the structure of ESN364, more commonly known as Veozah.
Its proper name is (R)-(4-fluorophenyl)-(8-methyl-3-(3-(methyl)-1,2,4-thiadiazol-5-yl)-5,6-dihydro-[1,2,4]trizolo[4,3-a]pyrazin-7(8H)-yl)methanone. Note that it is a Chiral molecule.
Deuterated form patented
Google Patents is another good place to look.
The original patent application5 was lodged in 2015, followed by a series of worldwide applications.
Here is a patent covering a tablet form for its protonated salts.6
I found a patent covering the use of the Deuterated drug7 that expires in 2038.
Deuterated fezolinetant increased the half-life by a factor 2 in castrated monkeys, compared to fezolinetant. Why were male monkeys used for a drug exclusvely intended for Women?
The patent covers an extraordinary list of alleged uses:
preventing depression, anxiety, psychosis, schizophrenia, psychotic disorders, bipolar disorders, cognitive disorders, Parkinson's disease, Alzheimer's disease, attention deficit hyperactivity disorder (ADHD), pain, convulsion, obesity, inflammatory diseases including irritable bowel syndrome (IBS) and inflammatory bowel disorders, emesis, pre-eclampsia, airway related diseases including chronic obstructive pulmonary disease, asthma, airway hyperresponsiveness, bronchoconstriction and cough, urinary incontinence, reproduction disorders, contraception and sex hormone-dependent diseases including but not limited to benign prostatic hyperplasia (BPH), prostatic hyperplasia, metastatic prostatic carcinoma, testicular cancer, breast cancer, ovarian cancer, androgen dependent acne, male pattern baldness, endometriosis, abnormal puberty, uterine fibrosis, uterine fibroid tumor, uterine leiomyoma, hormone-dependent cancers, hyperandrogenism, hirsutism, virilization, polycystic ovary syndrome (PCOS), premenstrual dysphoric disease (PMDD), HAIR-AN syndrome (hyperandrogenism, insulin resistance and acanthosis nigricans), ovarian hyperthecosis (HAIR-AN with hyperplasia of luteinized theca cells in ovarian stroma), other manifestations of high intraovarian androgen concentrations (e.g. follicular maturation arrest, atresia, anovulation, dysmenorrhea, dysfunctional uterine bleeding, infertility), androgen-producing tumor (virilizing ovarian tumor or virilizing adrenal tumor), menorrhagia and adenomyosis.
And then tags in:
Another object of the invention is (R)-(4-fluorophenyl)-(8-methyl-3-(3-(methyl-d3)-1,2,4-thiadiazol-5-yl)-5,6-dihydro-[1,2,4]trizolo[4,3-a]pyrazin-7(8H)-yl)methanone or a pharmaceutically acceptable salt or solvate thereof for use in treating and/or preventing hot flashes.
A further object of the invention is (R)-(4-fluorophenyl)-(8-methyl-3-(3-(methyl-d3)-1,2,4-thiadiazol-5-yl)-5,6-dihydro-[1,2,4]trizolo[4,3-a]pyrazin-7(8H)-yl)methanone or a pharmaceutically acceptable salt or solvate thereof for use as a lowering agent of the circulating LH levels.
Patent searching so far did not provide me with satisfactory information on metabolites of Veozah.
Fluoride from 4-Fluorophenol as an example of Clear and Present Danger
In 2012 it was reported that Cytochrome P450BM3-F87G catalyzes Defluorination of 4-Fluorophenol followed by reduction of the resulting benzoquinone to hydroquinone via the NADPH P450-reductase.8 Co-incubation of the reaction mixture with long chain aldehydes stimulated the Defluorination reaction.
This followed a 1997 report.9
Many useful papers are behind paywalls.1011
Defluorination of drugs is of great interest because many are Persistent Organic Pollutants and a recent paper showed that Iron Oxo centres can help liberate free Fluoride from F-substituted aromatic rings.12
Ultraviolet light liberates Free Fluoride from 4-Fluorophenol.13
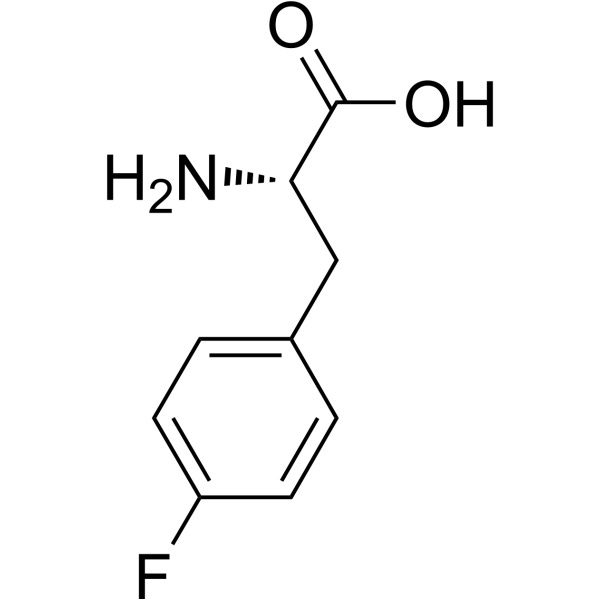
Fluoride from 4-Fluorophenyl-L-Phenylalanine as an example of Clear and Present Danger
This molecule was shown in 1961 to be defluorinated by Phenylalanine Hydroxylase to yield free Fluoride ion.14 It is also a substrate for Tyrosine Hydroxylase.
Not all paraFluoro aromatics are defluorinated in vivo, at least not in bone in one study.15
Defluorination of Veozah by Bacterial Enzymes in the Gut ?
An additional hazard that might help explain differences in individual risk from Fluorinated aromatic drugs, is the potential for Defluorination by the bacteria in their Guts. This can be affected by optical isomers. It has been reported that only the L-Leucine specific receptor of Escherichia coli binds 3-L-FluoroPhenylalanine and 4-L-FluoroPhenylalanine.16
Have you found Human metabolites of Veozah ?
Please let me know if you get lucky while I continue the search.
https://sharylattkisson.com/2024/09/read-fda-warning-added-hot-flash-drug-veozah/
https://ctdbase.org/detail.go?type=chem&acc=C000608808
Hamid Hoveyda, Guillaume Dutheuil, Graeme Fraser. 2015. Substituted [1,2,4]triazolo[4,3-a]pyrazines as selective NK-3 receptor antagonists. US9422299B2. https://patents.google.com/patent/US9422299B2/ru
Helena CERIĆ and Nikolina JANTON. 2021. Solid state forms of fezolinetant and salts thereof. WO2022031773A1. https://patents.google.com/patent/WO2022031773A1/en
Hamid Hoveyda. Assigned to Ogeda SA. 2018. Deuterated fezolinetant. US11078203B2. https://patents.google.com/patent/US11078203B2/en
Alexandria Harkey, Hye-Jin Kim, Suneel Kandagatla and Gregory M. Raner. 2012. Defluorination of 4-fluorophenol by Cytochrome P450BM3-F87G: Activation by long Chain Fatty Aldehydes. Biotechnology Letters. 34:1725-31
Osman AM, Boeren S, Veeger C, Rietjens IMCM. 1997. MP8-Dependent oxidative dehalogenation: evidence for the direct formation of 1,4-benzoquinone from 4-fluorophenol by a peroxidase-type of reaction pathway. Chem Biol Interact 104:147–164
Jiaxin Zhu, Zekun Liu, Yihua Chen, Zhiqiang Chen, Hang Ma, Jian Sun, Liao Meng, Tongzhou Liu. 2023. A detailed investigation of electrochemical hydrodefluorination of 4-fluorophenol: Rh-doped cathode preparation, reaction conditions optimizations, reactive species identification, and in situ characterization. https://www.sciencedirect.com/science/article/abs/pii/S1385894723027407
Yi Zhang, Thirakorn Mokkawes, Sam P. de Visser. 2023. Insights into Cytochrome P450 Enzyme Catalyzed Defluorination of Aromatic Fluorides. https://onlinelibrary.wiley.com/doi/10.1002/anie.202310785
Umesh Kumar Bagha, Dr. Rolly Yadav, Thirakorn Mokkawes, Jagnyesh Kumar Satpathy, Prof. Devesh Kumar, Prof. Chivukula V. Sastri, Dr. Sam P. de Visser. 2023. Defluorination of Fluorophenols by a Nonheme Iron(IV)-Oxo Species: Observation of a New Intermediate Along the Reaction. https://chemistry-europe.onlinelibrary.wiley.com/doi/full/10.1002/chem.202300478
Laxiang Yang, Jieqiong Zhou and Yuxin Feng. 2021. Treatment of fluorine-containing pharmaceutical wastewater by VUV/UV process. https://link.springer.com/article/10.1007/s11356-021-17063-8
S. Kaufman. Hydroxylation of 4-fluoro-L-phenylalanine by phenylalanine hydroxylase results in L-tyrosine and fluoride ion. Biophysica et Biochimica Acta 19, 619-627 (1961).
Sang Hee Lee, Nunzio Denora, Valentino Laquintana, Giuseppe Felice Mangiatordi, Angela Lopedota, Antonio Lopalco, Annalisa Cutrignelli, Massimo Franco, Pietro Delre, In Ho Song, Hye Won Kim, Su Bin Kim, Hyun Soo Park, Kyungmin Kim, Seok-Yong Lee, Hyewon Youn, Byung Chul Lee, and Sang Eun Kim. Radiosynthesis and characterization of [18F]BS224: a next-generation TSPO PET ligand insensitive to the rs6971 polymorphism. 2021. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8712300/
Linda A. Luck, Colin Johnson. 2008. For the record: Fluorescence and 19F NMR evidence that phenylalanine, 3-L-fluorophenylalanine and 4-L-fluorophenylalanine bind to the L-leucine specific receptor of Escherichia coli. https://onlinelibrary.wiley.com/doi/10.1110/ps.9.12.2573