In October 2022, the UK Government issued an urgent recall1 of Targocid due to Endotoxin contamination.
It is Teicoplanin, a “semisynthetic” glycopeptide antibiotic.2
Another trade name is Ticocin marketed by Cipla (India).
Teicoplanin has demonstrated in vitro efficacy against Gram-positive bacteria including staphylococci (including MRSA), streptococci, enterococci, and against anaerobic Gram-positive bacteria including Clostridium spp.
Teicoplanin is not effective against Gram-negative bacteria
Australia TGA very worried in 1993
In 1993 the Therapetic Goods Administration told Sanofi Aventis via MERRELL DOW PHARMACEUTICALS it was not satisfied with their submission because of the Water for Injection Endotoxin contamination.3 Look at this:
Click the image or read my snippets.
The Company has provided a method and results for the Bacterial Endotoxin Test on the Water for Injection supplied with the TARGOCID powder (Bl App 4).
No Endotoxin limit is given (the FDA Guidelines 1990 list a 0.25EU/ml limit);
the method does not comply with the USP XXII 1990 <85> Bacterial Endotoxins Test and the proposed revisions in the Pharmacopial Forum Sept-Oct 1992 Vol 18, No 5.
This is unsatisfactory.
The method needs to include the following:
a) Calibration of a CSE with the RSE;
b) Confirmation of the labeled LAL reagent sensitivity;
c) Inhibition/Enhancement test with CSE standard curves in water and product;
d) Maximum Valid Dilution;
e) Test procedure with a CSE standard curve in water; and
f) Confirmation that the supplier of reagents is approved by the FDA.
Note the deference to US FDA LIMIT of Endotoxin contamination to 0.25 EU/ml and compare that with Kevin McKernan measurement of 19 EU/ml in a batch of Pfizer Jab.
TARGOCID, marketed by Sanofi Aventis is a mixture of 9 different compounds, some of which have chiral side chains (remember Thalidomide and LNP ALC-0315).
How Much Endotoxin in TARGOCID ??
The released recall notice for 2 batches simply says “out of specification results obtained for bacterial endotoxins, which has been confirmed through testing of retain samples.”
Please let me know if you discover the actual Endotoxin measured in EU/ml.
Advice for all healthcare professionals:
Healthcare professionals should be aware of the following clinical symptoms related to the potential risk to patient health:
High temperature (Fever) or
Low body temperature, Chills and Shivering
Cold, Clammy and Pale or Mottled skin
Fast Heartbeat
Fast Breathing
Severe Breathlessness
Severe Muscle Pain,
Feeling Dizzy or Faint,
Change in Mental State – such as Confusion or Disorientation
Loss of Consciousness
Slurred Speech
Nausea and
Vomiting and/or Diarrhoea.
Readers will immediately relate these to the most common Adverse Events of Jabbing with any product containing Endotoxin.
Thanks to Chris Edwards who alerted me to this UK recall which was done while I was suspended from Twitter (now X).
I had downloaded the TGA FOI some months ago and saved it for a rainy day.
Injecting antibacterials can release Endotoxin
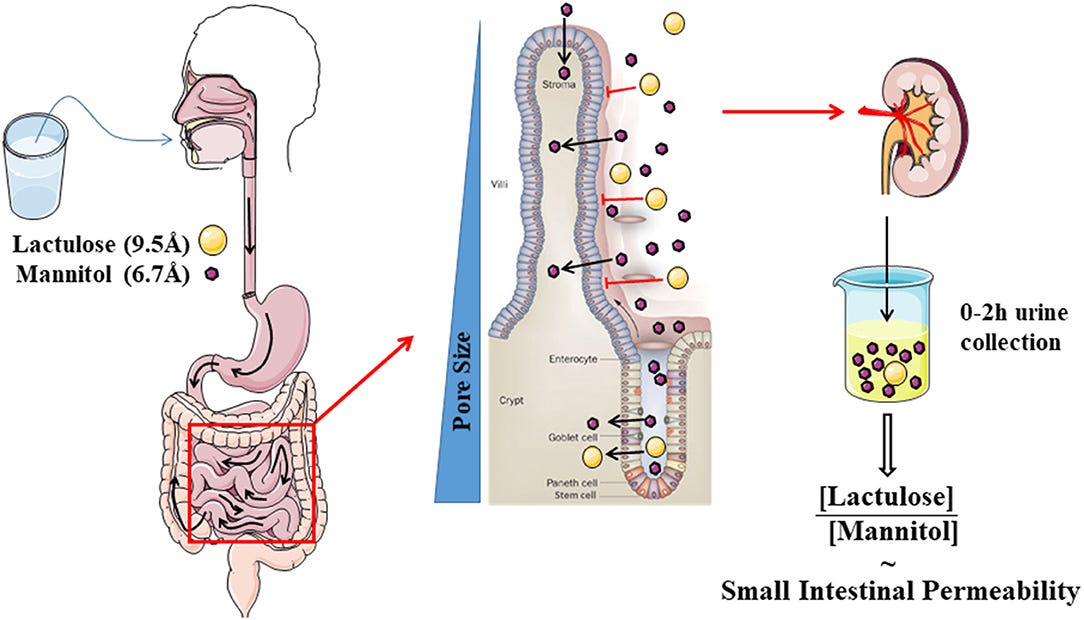
A note of caution that products like Targocil or other Endotoxin contaminated Jabs can do more harm than good by opening the Gut4, killing the Bacteria and releasing even more Endotoxin.5
https://www.gov.uk/drug-device-alerts/national-patient-safety-alert-class-1-medicines-recall-notification-recall-of-targocid-200mg-powder-for-solution-for-injection-slash-infusion-or-oral-solution-aventis-pharma-limited-t-slash-a-sanofi-due-to-the-presence-of-bacteria
https://en.wikipedia.org/wiki/Teicoplanin
TGA. 1993 FOI 131-1617 Document 9.
Daniele Focosi. Antimcrobials for Bacteria. https://www.ufrgs.br/imunovet/molecular_immunology/antimicrobialsbacteria.html